Submitting Physical Specimens for Diagnosis
Please fill out the Physical Sample Submission Form (PPDL-Form-006-004) and include with your sample.
Contact Us
Plant and Pest Diagnostic Laboratory
Purdue University
915 Mitch Daniels Boulevard, LSPS 116
West Lafayette IN 47907-2054
Email: ppdl-samples@purdue.edu
Voice: 765-494-7071
How to Collect and Ship Specimens
- Collect fresh specimens. Send a generous amount of material, if available.
- Ship in crush-proof container immediately after collecting. If holdover periods are encountered, keep specimen cool. Mail packages to arrive on weekdays. Do not mail or ship samples on Friday, as we are not here to receive them over the weekend. We encourage you to send samples with priority or express delivery so we receive them in the best condition possible to provide you with the most accurate diagnosis.
- Incomplete information or poorly selected specimens may result in an inaccurate diagnosis. Badly damaged specimens are often unidentifiable and additional sample requests can cause delays.
- Please double bag out-of-state samples to prevent escape of pathogens/pests during shipping.
Mailing your sample: We are not open on the weekend. Ship early in the week (Monday-Wednesday) using a next day deliver option to make sure your sample gets to us before the weekend. For Thursday shipments DO NOT use USPS overnight delivery. Your package may arrive at Purdue’s central receiving dock by Friday but will not get to our lab by close of business on Friday. Instead use only UPS or FedEx if shipping on Thursday.
NEMATODE SAMPLES
Nematode services we provide:
- Soybean cyst nematodes in roots
- Root knot nematode in root samples
- Foliar nematodes in leaf samples
- Beach leaf disease nematodes in leaf samples
Our lab does not do nematode counts from soil samples. For a list of nematode labs in the region that do soil samples please contact the PPDL, ppdl-samples@purdue.edu or 765-494-7071.
Submitting Plant Specimens for Disease/Injury Diagnosis
Herbaceous Plants
For general decline/dying of plants, send whole plants, showing early symptoms, with roots and adjacent soil intact. Dig up plant carefully. Send several plants. Bundle plants together and wrap roots in a plastic bag. Wrap the entire bundle of plants in newspaper and place in a crush-proof container for shipment. Do not add water.
Tree Wilts
Collect branches 1/2 to 1 inch in diameter from branches which are actively wilting but not totally dead. Wrap in plastic to retain moisture. Collect a handful of feeder roots and place in a plastic bag. Oak wilt samples require special handling and should be shipped in a cooler with an ice pack included. Be sure to insulate the ice pack with bubble-wrap or other material so the cooler stays cool but the plant parts do not sit directly against the ice pack. Overnight shipment is especially important for oak wilt samples.
Leaves/Branches/Fleshy Parts
When localized infections such as cankers, leaf spots and rots are involved, send specimens representing early and moderate stages of disease. For cankers include healthy portions from above and below diseased area. Press leaves flat between heavy paper or cardboard. Wrap fleshy parts in dry paper.
Turf
Samples should be at least 4"x4" and include both the diseased and healthy portions of grass on the same sample piece. Place the sample on aluminum foil and wrap tightly to keep soil off the blades of grass. Please use PPDL-Form-006-005 for all turf submissions.
Click image to enlarge
Submitting Plant Specimens for Identification
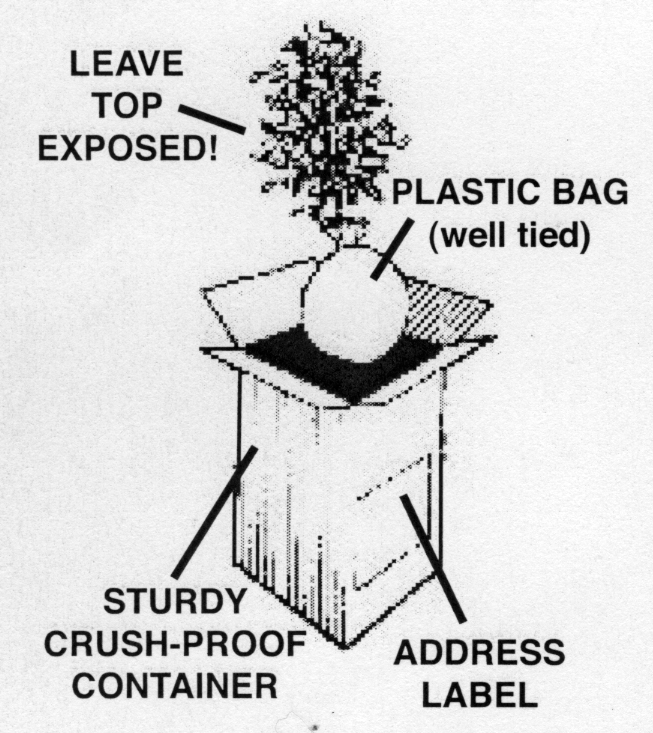
- Include a 6-10 inch sample of the terminal (tip) portion of the stem with side buds, leaves and flowers in identifiable condition.
- Place the sample flat between a layer or two of dry newspaper, paper toweling or similar absorbent material. Try to prevent excessive folding of the leaves and place flowers so that you are looking into the center of the flower.
- Pack the wrapped bundle in plastic, preferably with a piece of cardboard to keep the sample flat.
- Never place any fresh plant sample directly in plastic.
- Never add water to the sample.
- Shake excess water from aquatic weed samples and place in plastic bag.
- Wrap whole, uncut fruit specimens in paper, place in a strong box, and pack with additional paper to prevent crushing.
- Package in sturdy crush-proof container and pack with additional paper to prevent shifting. See above photo.
- The PPDL no longer has a faculty member working on algae and aquatic weeds. Please contact for a list of aquatic service companies for assistance with control.
Submitting Insect Specimens
Samples related to health problems (suspected biting insects)
NOTE: This type of sample must be submitted in a small leak-proof vial/bottle in 70% alcohol (isopropyl). The sample must contain only the insect, nothing else. We do not accept articles of clothing or sticky traps for insect identification. We cannot diagnose signs or symptoms on people or animals from photos.
Tiny and/or Soft-bodied Specimens
Aphids, mites, thrips, caterpillars, grubs, spiders, etc. should be submitted in a small leak-proof bottle or vial of 70 percent alcohol. Rubbing (isopropyl) alcohol is suitable and readily available. Do not submit insects in water, formaldehyde or without alcohol as they will readily ferment and decompose.
*Grubs and caterpillars must be prepared before preservation to prevent discoloration. Drop them into gently boiling water for about 30 seconds (never microwave them!) before placing in vials of rubbing alcohol.
Hard-bodied Specimens
Flies, grasshoppers, cockroaches, wasps, butterflies and beetles can be submitted dry in a crush-proof container. Do not tape insects to paper or place them loose in envelopes.
Submitting Greenhouse Samples
Samples in plug trays, as well as unrooted and rooted cuttings, and plants in pots require extra care when they are packaged for submittal to a diagnostic lab. Before you mail the next sample, please take a few minutes to review these suggestions for packaging and submitting samples. This will help preserve the integrity of the sample during shipment and increasing the likelihood of a more accurate diagnosis.Plugs
If possible, do not remove the plugs from the plug tray. Submitting either an entire tray or cutting off a section of the tray helps keep the soil off the foliage where most symptoms are observed. Secondary decay often occurs when soil is allowed to come in contact with the foliage, interfering with accurate diagnosis. When possible, submit at least 5-10 cells with plugs. This provides the diagnostician with ample material for microscopic observation, culturing, and virus testing if necessary.
Cuttings
The primary concern is to keep the growing media separate from the foliage. You would be amazed how quickly damp foliage with a dusting of growing mix rots once it is sealed in packing material! Put the cuttings into a plastic bag, sealing the bag with a twist tie at the soil line. Do not seal the foliage in a plastic bag. Then wrap the sample in newspaper to prevent additional drying out of foliage before it is received.
Potted Material
Take into consideration that the mail carrier will not necessarily keep these packages right side up. Wrap plastic wrap, clear packing tape or paper over the pot surface, or put the pot in a bag and seal it with a twist tie around the base of the plant. Fill any extra space in the shipping box with newspaper, Styrofoam peanuts, or another space filling packing material to prevent jostling of sample during shipment.